How to Verify Botanical Extract Authenticity by 2028?
May 09, 2026
Leave a message
How to Verify Botanical Extract Authenticity by 2028?
Strategic Whitepaper by Xi'an Tihealth R&D Division | Forensic Phytochemistry & Compliance
Trust in the global botanical extract market is collapsing. By 2028, a simple HPLC purity claim will not survive a top-tier pharmaceutical audit. Synthetic biology has flooded the supply chain. Cheap, petroleum-derived molecules easily masquerade as "natural-equivalent" extracts. At Xi'an Tihealth, we believe that in modern B2B procurement, Forensic Data Integrity is the only metric that matters. Purchasing decisions are no longer driven by price tags; they are dictated by isotope signatures and DNA barcoding. Let us strip away the marketing fluff and dissect the brutal physicochemical realities governing high-performance botanical bioactives.
Why Is Standardization Superior to Ratio Extracts?
The "10:1 Extract" label is a massive regulatory loophole. Unquantified ratio extracts are chaotic variables in pharmaceutical engineering. Because raw biomass fluctuates based on geography, harvest season, and drying protocols, the secondary metabolite profile of a ratio extract can vary by up to 400% between batches. You cannot formulate a clinical-grade product with moving targets.
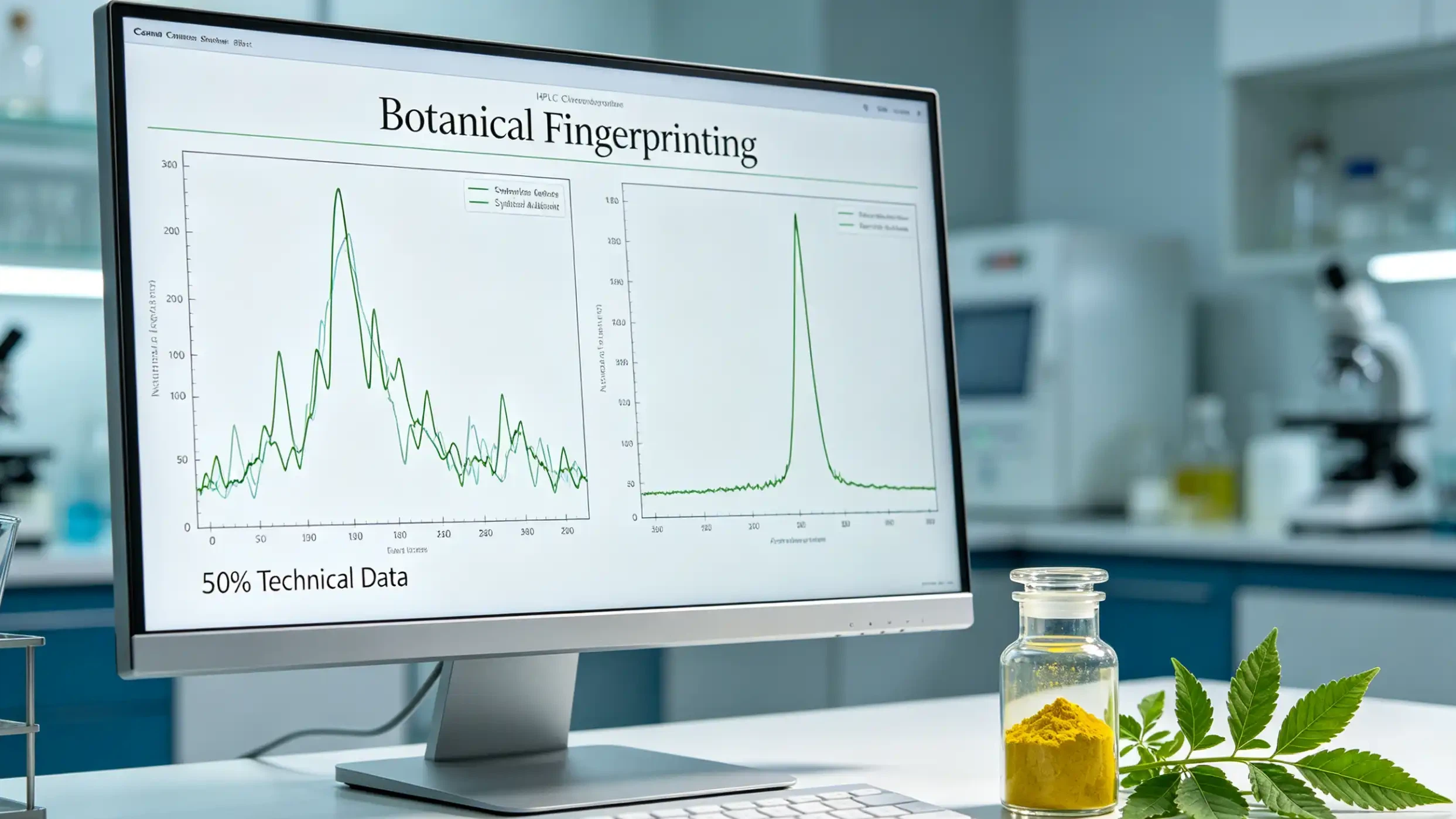
Forensic HPLC profiling distinguishing natural botanical fractions from synthetic spiked adulterants.
Xi'an Tihealth strictly enforces Standardized Extraction. We deploy precise HPLC and UV-Vis quantification for core bioactives (like Resveratrol, Apigenin, and Curcumin). Furthermore, we map the entire chemical matrix. By enforcing "Full Fingerprint Consistency," we guarantee that the synergistic secondary compounds remain identical batch after batch. In the 2028 regulatory climate, suppliers failing to provide comprehensive chromatographic maps will be purged from high-value supply chains.
How to Detect Synthetic Adulteration Using 14C Testing?
This is the ultimate anti-fraud mechanism. Synthetic molecules derived from fossil fuels contain zero Carbon-14 (14C). Conversely, genuine botanical extracts, harvested from living plants that breathe atmospheric CO2, possess a highly specific 14C isotopic signature.

Carbon-14 isotopic radiocarbon dating to prove 100% natural botanical origin.
By utilizing Accelerator Mass Spectrometry (AMS), we can instantly differentiate between a bio-based extract and a cheap, synthetic lab clone. For easily adulterated, high-value molecules, Xi'an Tihealth proactively supplies third-party 14C isotopic verification. This absolute physical proof is a non-negotiable clause in premium 2028 procurement contracts. Without this data, "100% Natural" is just empty marketing.
Does Your Extract Meet USP <561> Pesticide Mandates?
Agrochemical residues and heavy metals are silent killers in botanical sourcing. As the European EP 2.8.13 and the American USP <561> frameworks aggressively tighten their limits, legacy extraction techniques are failing outright. Plants act as bio-accumulators. They absorb heavy metals (Lead, Mercury, Arsenic, Cadmium) directly from the soil.
We deploy advanced Ion-Exchange Resins and nano-filtration systems to strip heavy metals down to strict parts-per-billion (ppb) thresholds. Regarding solvent residues, Xi'an Tihealth is transitioning our core premium lines to Supercritical CO2 (S-CO2) Extraction for 2028. This cryogenic, oxygen-free process guarantees absolute zero solvent residue. It protects highly volatile terpenoids and strictly adheres to the global "Clean Label" mandate.
Why is DNA Barcoding Mandatory for Raw Biomass?
Macroscopic visual identification is obsolete. Unscrupulous brokers routinely substitute high-value botanicals (like Panax ginseng) with closely related, visually identical, yet pharmacologically inert generic variants.

Genomic DNA barcoding to prevent species adulteration in raw botanical sourcing.
DNA barcoding provides exact genomic species identification at the point of origin. By authenticating the raw material before it ever enters the extraction vessel, Xi'an Tihealth eliminates the risk of species adulteration, ensuring the safety and efficacy of the final botanical matrix.
High-Tier Botanical TDS Benchmarks (2028)
| Quality Variable | Standard Industry Practice | Xi'an Tihealth Precision Standard |
|---|---|---|
| Purity Verification | Single peak HPLC | Fingerprint Matching & 14C Isotope |
| Solvent Residue | Acetone/Hexane < 5000ppm | S-CO2 Extraction (Zero Solvent) |
| Heavy Metals | < 10ppm total screening | Pb < 0.5ppm / As < 0.1ppm (ICP-MS) |
| Particle Size | Basic 80 Mesh powder | D90 < 10μm (Micronization available) |
| Stability | Standard poly-bag packaging | Nitrogen-purged Vacuum Sealing |
Strategic Botanical Extracts Sourcing FAQ
1. What exactly is a Botanical Fingerprint?Regulatory References & Technical Standards
- USP <561>: Articles of Botanical Origin - Pesticide Residue Analysis. Official Guideline
- EP 2.8.13: European Pharmacopoeia - Pesticide residues in herbal drugs.
- ICH Q3C: Impurities: Guideline for Residual Solvents.
- ASTM D6866: Standard Test Methods for Determining the Biobased Content of Samples using Radiocarbon Analysis.
Send Inquiry






